 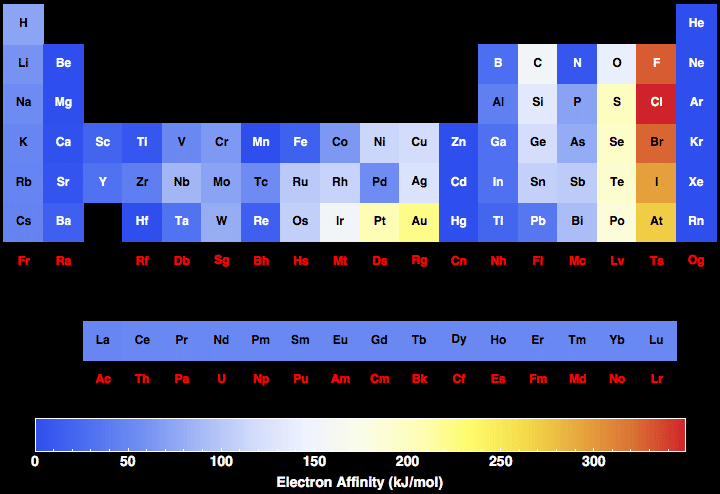
This results in a higher electronegativity value for Beryllium, which has a value of 1.6 compared to 1.0 for Lithium. Out of these 118, only 94 are naturally occurring. And the element which has the lowest electronegativity value is Francium in 0.7 χ. How did hubbard arrange the elements Last Update: May 30, 2022. The element which has the highest electronegativity value is Fluorine with 3.98 χ. in fact order most elements according to trends in their atomic weights. You can print the list of elements by hitting the print button below. There is a sense in which the chemical properties can be regarded as more basic. To list the elements order by electronegativity, click on the table headers. List of elements ordered by electronegativity is listed in the table below with atomic number, chemical symbol and electronegativity value. 
What are the group number and the generic outer electron configuration for a neutral atom with the given Lewis symbol ? Review images B.List of Elements in Order of Electronegativity χ Which element could this symbol represent? Ge A. 
8 Consider the generic Lewis dot symbol for an element. How many valence electrons are in the Lewis-dot (electron dot) structure for the neutral krypton (Kr) atom? A. How many valence electrons are in the Lewis-dot (electron dot) structure for the neutral silicon (Si) atom?ī. Ne Which of these elements has the highest first ionization energy? chlorine Complete the Lewis dot (electron‑dot) structure for the given neutral atoms. *refer to image* Largest - smallest radius Li Arrange the elements according to atomic radius. *refer to image* Largest - smallest radius: Rb Arrange these elements according to atomic radius. *refer to image* Most - least electronegative: The periodic table is arranged according to the periodic law. f Arrange these elements according to electronegativity. Similar to atomic radius, the ionic radius of an element increases in size down a group as. Refer to diagram Label these blocks of the periodic table according to the highest‑energy occupied subshell for the elements. Use proper formatting letter case matters. Refer to diagram Determine the chemical symbols for the neutral elements corresponding to the electronic configurations. What is the atomic symbol for the noble gas that also has this electron configuration? 1s^2 2s^2 2p^6 3s^2 3p^6Īrgon Group the electronic configurations of neutral elements in sets according to those you would expect to show similar chemical properties. 2s^2 2p^2 Write the full electron configuration for P^3− 1s^2 2s^2 2p^6 3s^1 Complete the electron configuration for C. Correct option is A) As we move from top to bottom in a group the electronegativity ( measure of the tendency of an atom to attract a bonding pair of electrons) will be decreased because the atomic radius will be increased so distance from outer electrons to the nucleus will be increased, so the electrons cannot be attracted easily. 6 Give the full electron configuration for sodium (Na). 
Argon Determine the number of valence electrons in each of the neutral atoms.Į. N = 4 - (2) What is the neutral atom that has its first two energy levels filled, has 88 electrons in its third energy level, and has no other electrons? Enter the name of the element, not the abbreviation. If the energy level does not contain any electrons, enter a 0. d Refer to image Enter the number of electrons in each energy level (shell) for each of the elements. 2 Classify the atomic orbitals as s, p, or d according to their shape.ģ. 16 Number of electrons to fill each sublevel:ĭ. Group of answer choices Ionization energy Electronegativity Atomic radius None. How many orbitals are in the □=4n=4 shell? A. Solution for Arrange the following ions in order of increasing ionic radius: calcium ion, potassium ion. How many subshells are in the □=2 shell?ī. What is the maximum number of electrons in the □=3 level? A. How many orbitals are in the □=3 level?Ĭ. How many sublevels are in the □=3 level?ī. If, at a given time, the energy of a subatomic particle is precisely known, then The time over which the measurement was made cannot be precisely known. What happens when an electron jumps from energy level 4 to energy level 2 in an atom?Ĭase 2: An electron jumps from energy level 4 to energy level 1 in an atom.Īssuming that both cases describe Hydrogen‑like atoms with one electron, for which case is more energy emitted or absorbed? Case 1: A photon is emitted by the atom.Ĭase 2: More energy is emitted or absorbed for case 2. 6 Case 1: An electron jumps from energy level 4 to energy level 2 in an atom. Using the image, determine how many different electronic transitions are possible when electrons fall from the fourth energy level (□=4)(n=4) to the first energy level (□=1)(n=1). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
